UConn SHaW is excited to introduce the new Wellness to Go, over-the-counter (OTC) vending machine! Conveniently located on the main floor of the Student Union, across from the information desk and next to the One Card office, this vending machine offers easy access to many of our most popular OTC products. Available exclusively to UConn undergraduate and graduate students who have paid the Storrs Campus Wellness Fee. Purchases can only be made using HuskyBucks.
Don’t see something you need in the vending machine? Order on our online marketplace for your over the counter needs!
SHaW also carries Plan B for free (undergrads only) in our Pharmacy during business hours.
Google Maps

Product Information:
AccuHome Pregnancy Test
Front of package

Side of package



Back of package

Medication Use Label Information: Germaine Laboratories, Inc.
Manufacturer: Germaine Laboratories, Inc.; Product ID: 89101 UPC: 695684891015
Acetaminophen Caplets (Brand Name: Tylenol®)
Front of package
Drug Facts



Medication Use Label Information: Acetaminophen
Manufacturer: MAJOR Pharmaceuticals; NDC: 0904-6720-24
Anti-Diarrheal loperamide caplets (Brand Name: Imodium®)
Front of package

Back of package

Medication Use Label Information: Loperamide
Manufacturer: Amerisource Bergen, LLC; NDC: 46122-0738-53
Artificial Tears (Geri Eye Drops – Dry Eye Relief)
Front of package

Back of package

Side of Package

Medication Use Label Information: ARTIFICIAL TEARS- glycerin solution/ drops
Manufacturer: Geri-Care Pharmaceuticals, Corp; NDC: 57896-181-05
Ayr® Saline Nasal Gel
Front of package
Back of package

Medication Use Label Information: Ayr Saline Nasal Gel
Manufacturer: B.F. Ascher & Co., Inc.; NDC: 00225-0525-47
Bacitracin Zinc Ointment
Front of package
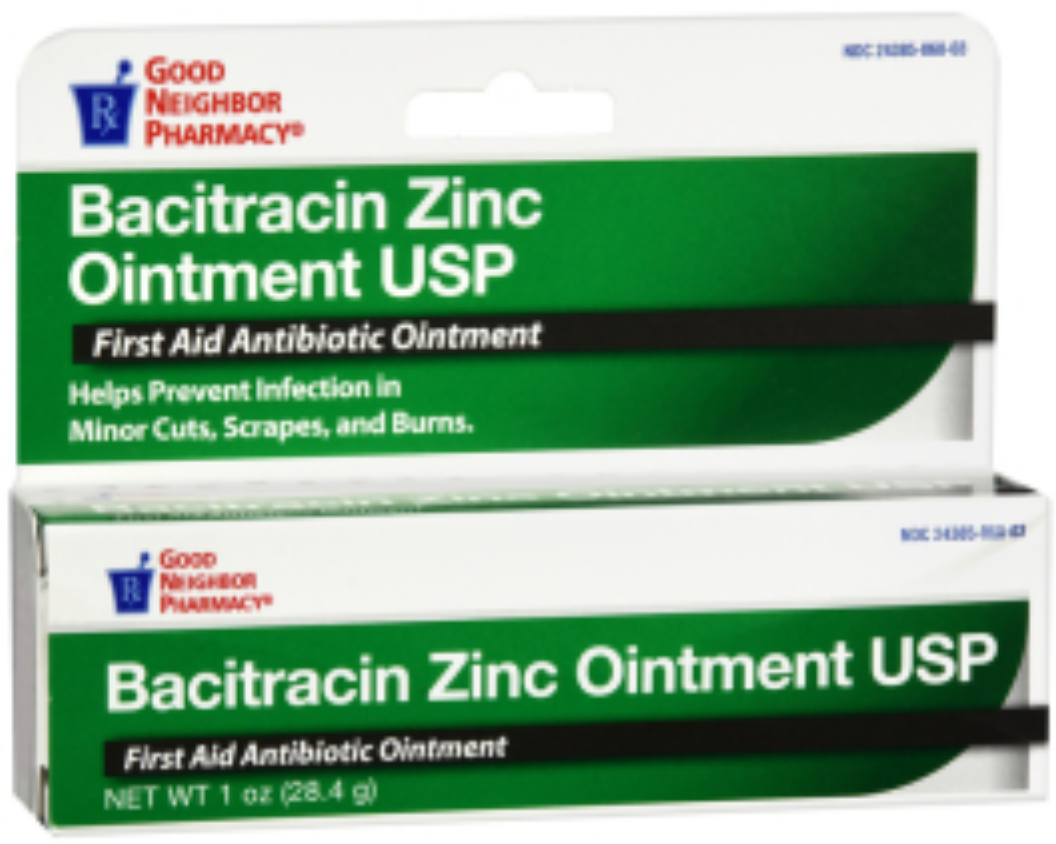
Back of package

Medication Use Label Information: Bacitracin zinc ointment
Manufacturer: Amerisource Bergen; NDC: 24385-060-03
Bandaids
Front of package


Medication Use Label Information: Bandaids
Calcium Carbonate Tablets (Brand Name: TUMS®)
Carefree® Panty Liners (pads)
Front of package

Back of package

Side of Package

Carmex® Classic Medicated Lip Balm
Front of package

Back of package

Medication Use Label Information: Carmex® Classic Medicated Lip Balm
Manufacturer: Carma Laboratories, Inc.; NDC: 0210-0023-1, 10210-0023-2, 10210-0023-3, 10210-0023-4, 10210-0023-5
Cold Max Day & Night (acetaminophen, phenylephrine hcl, dextromethorphan hbr, chlorpheniramine maleate)
Front of package
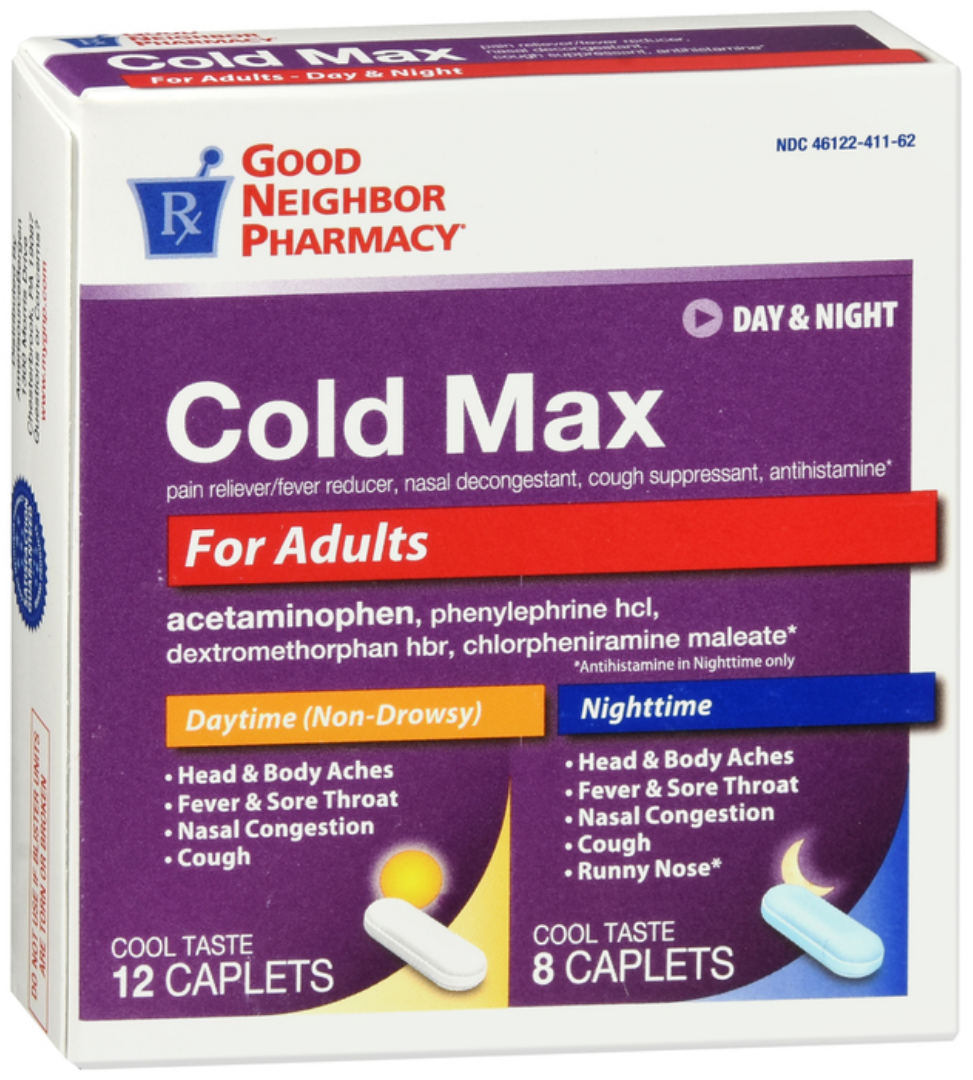
Back of package

Medication Use Label Information: Cold Max Day and Night
Manufacturer: AmeriSource Bergen; NDC: 46122-411-62
Colgate® Toothpaste
Front of package


Medication Use Label Information: COLGATE GREAT REGULAR FLAVOR
Manufacturer: Colgate-Palmolive Company
COVID-19 and Flu A/B Home Test
Front of package
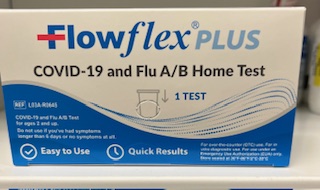
Back of package

Side of Package




Cough Drops (Menthol lozenge)
Front of package

Back of package

Medication Use Label Information: GOODSENSE MENTHOL COUGH DROPS- menthol lozenge
Manufacturer: Geiss, Destin, Dunn; NDC: 50804-013-30
Ibuprofen tablets (Brand Names: Advil®, Motrin®)
Front of package
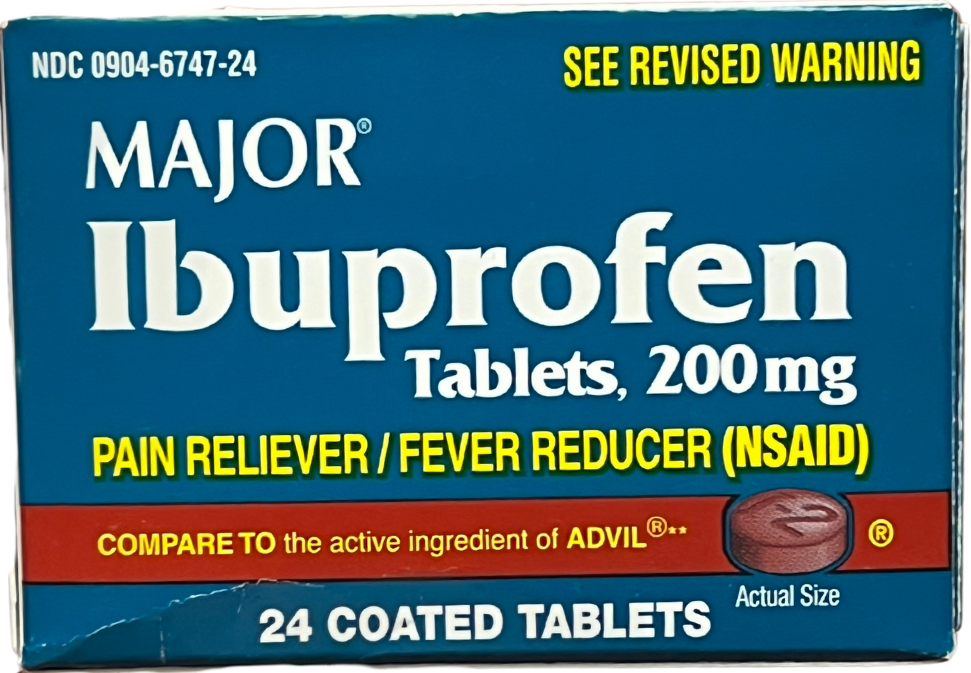
Back of package

Medication Use Label Information: Ibuprofen
Manufacturer: MAJOR Pharmaceuticals; NDC: 0904-6747-24
Loratadine tablets (Brand Name: Claritin®)
Front of package

Back of package

Medication Use Label Information: Loratadine tablet
Manufacturer: Bayer HealthCare LLC; NDC: 11523-7160-01
Miconazole Nitrate Vaginal Antifungal (Brand Name: Monistat 3®)
Front of package
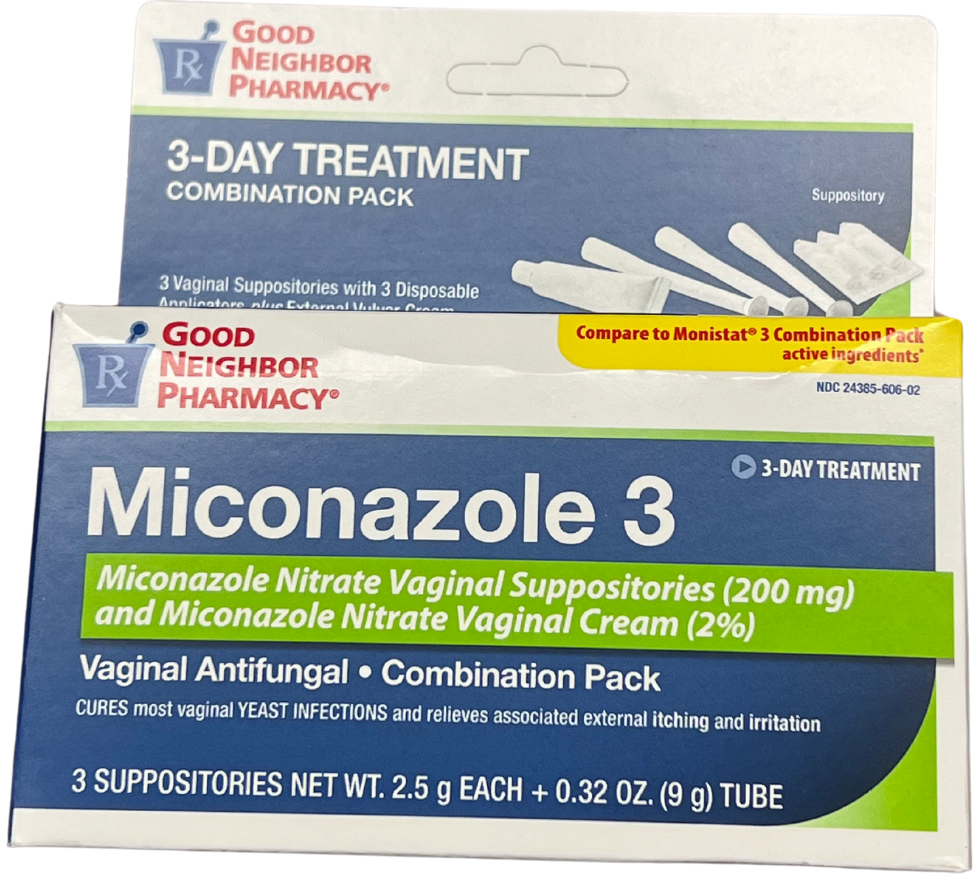
Back of package

Medication Use Label Information: Miconazole Nitrate
Manufacturer: Amerisource Bergen; NDC: 24385-0606-02
Mucinex® DM (dextromethorphan hydrobromide and guaifenesin tablet, extended release tablets)
Front of package

Back of package

Medication Use Label Information: Mucinex
Manufacturer: RB Health (US) LLC; NDC: 63824-072-46
Mucinex® ER (guaifenesin extended-release tablets)
Front of package

Back of package

Medication Use Label Information: GUAIFENESIN tablet, extended release
Manufacturer: AMERISOURCE BERGEN; NDC: 46122-750-51
My Way® Emergency Contraception (Levonorgestrel tablet)
Front of package

Back of package
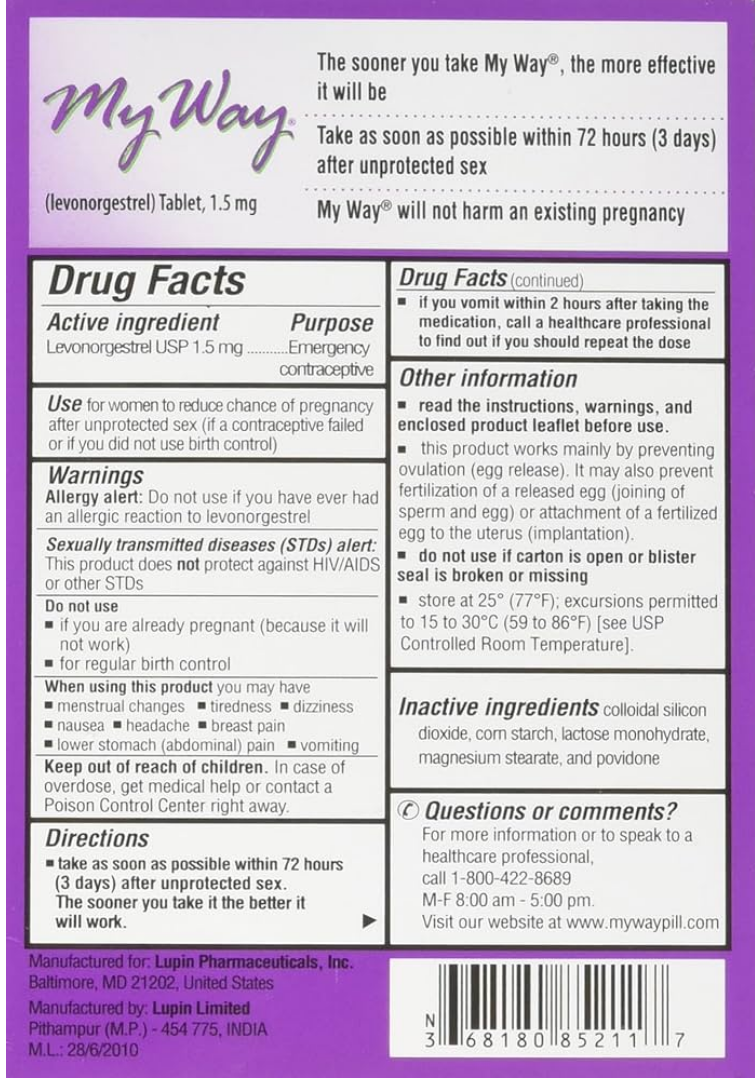
Medication Use Label Information: MY WAY- levonorgestrel tablet
Manufacturer: Lupin Pharmaceuticals, Inc.; NDC: 68180-852-11
Narcan® (Naloxone HCI Nasal Spray)
Front of package
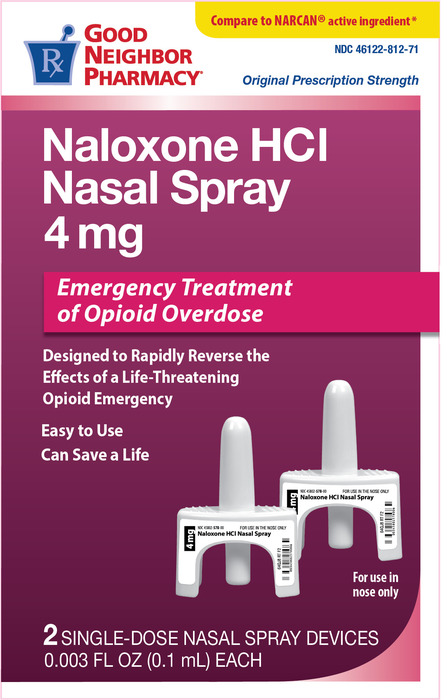
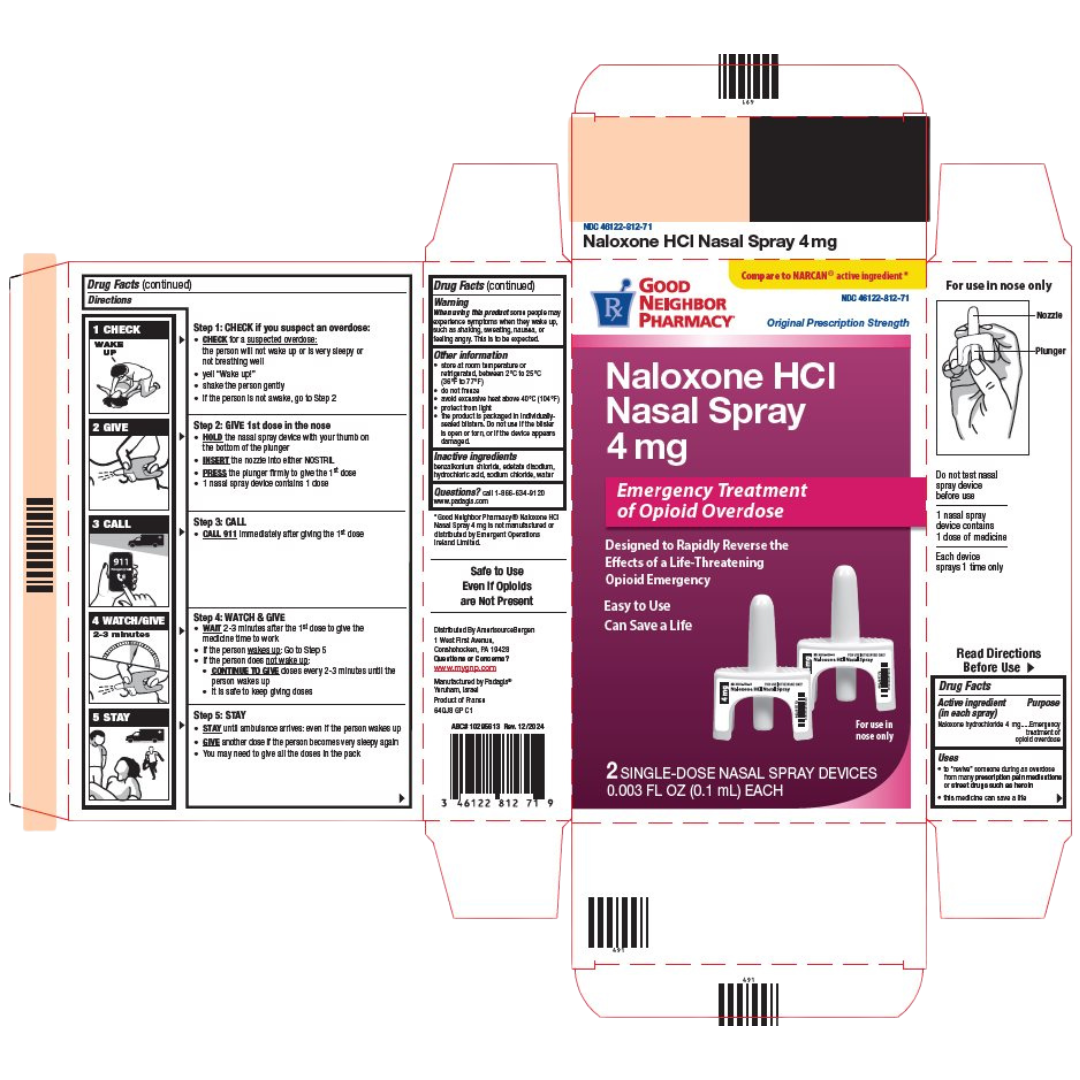
Medication Use Label Information: Narcan
Manufacturer: Padagis; NDC is 46122-0812-71
Tampax®(Tampons, unscented, super)
Front of package

Back of package

Product Use Label Information: Tampax Tampons
Manufacturer: Procter and Gamble; NDC: N/A
Trojan® ENZ Lubricated Latex Condoms 3 ct.
Front of package

Back of package

Product Use Label Information: Trojan Enz Lubricated Latex Condoms
Manufacturer: Church & Dwight Company
Urinary Pain Relief (phenazopyridine tablets) (Brand Name: AZO®)
Front of package

Back of package

Medication Use Label Information: Phenazopyridine
Manufacturer: AmerisourceBergen Drug Corp; NDC: 46122-628-53
Resources: